Page 153 - LAPORAN TAHUNAN HOSPITAL SULTANAH AMINAH 2022
P. 153
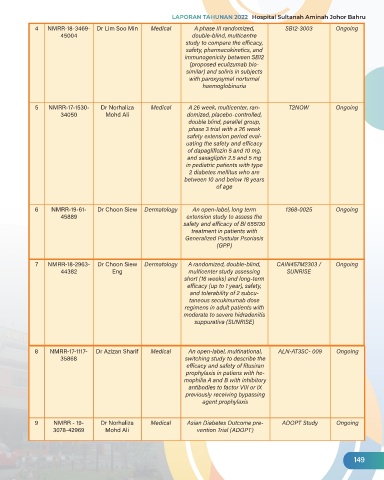
4 NMRR-18-3469- Dr Lim Soo Min Medical A phase III randomized, SB12-3003 Ongoing
45004 double-blind, multicentre
study to compare the efficacy,
safety, pharmacokinetics, and
immunogenicity between SB12
(proposed eculizumab bio-
similar) and soliris in subjects
with paroxysymal norturnal
haemoglobinuria
5 NMRR-17-1530- Dr Norhaliza Medical A 26 week, multicenter, ran- T2NOW Ongoing
34050 Mohd Ali domized, placebo-controlled,
double blind, parallel group,
phase 3 trial with a 26 week
safety extension period eval-
uating the safety and efficacy
of dapagliflozin 5 and 10 mg,
and saxagliptin 2.5 and 5 mg
in pediatric patients with type
2 diabetes mellitus who are
between 10 and below 18 years
of age
6 NMRR-19-61- Dr Choon Siew Dermatology An open-label, long term 1368-0025 Ongoing
45889 extension study to assess the
safety and efficacy of BI 655130
treatment in patients with
Generalized Pustular Psoriasis
(GPP)
7 NMRR-18-2963- Dr Choon Siew Dermatology A randomized, double-blind, CAIN457M2303 / Ongoing
44382 Eng multicenter study assessing SUNRISE
short (16 weeks) and long-term
efficacy (up to 1 year), safety,
and tolerability of 2 subcu-
taneous secukinumab dose
regimens in adult patients with
moderate to severe hidradenitis
suppurativa (SUNRISE)
8 NMRR-17-1117- Dr Azizan Sharif Medical An open-label, multinational, ALN-AT3SC- 009 Ongoing
35868 switching study to describe the
efficacy and safety of fitusiran
prophylaxis in patiens with he-
mophilia A and B with inhibitory
antibodies to factor VIII or IX
previously receiving bypassing
agent prophylaxis
9 NMRR - 19- Dr Norhaliza Medical Asian Diabetes Outcome pre- ADOPT Study Ongoing
3078-42969 Mohd Ali vention Trial (ADOPT)
149