Page 155 - LAPORAN TAHUNAN HOSPITAL SULTANAH AMINAH 2022
P. 155
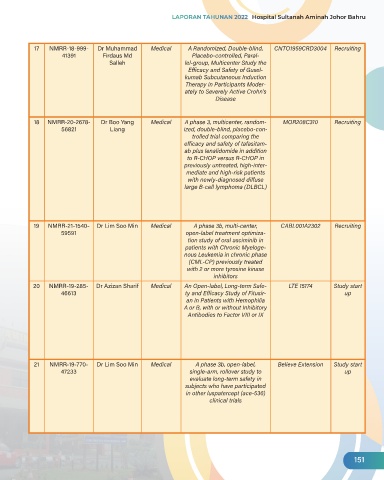
17 NMRR-18-999- Dr Muhammad Medical A Randomized, Double-blind, CNTO1959CRD3004 Recruiting
41391 Firdaus Md Placebo-controlled, Paral-
Salleh lel-group, Multicenter Study the
Efficacy and Safety of Gusel-
kumab Subcutaneous Induction
Therapy in Participants Moder-
ately to Severely Active Crohn's
Disease
18 NMRR-20-2678- Dr Boo Yang Medical A phase 3, multicenter, random- MOR208C310 Recruiting
56821 Liang ized, double-blind, placebo-con-
trolled trial comparing the
efficacy and safety of tafasitam-
ab plus lenalidomide in addition
to R-CHOP versus R-CHOP in
previously untreated, high-inter-
mediate and high-risk patients
with newly-diagnosed diffuse
large B-cell lymphoma (DLBCL)
19 NMRR-21-1540- Dr Lim Soo Min Medical A phase 3b, multi-center, CABL001A2302 Recruiting
59591 open-label treatment optimiza-
tion study of oral asciminib in
patients with Chronic Myeloge-
nous Leukemia in chronic phase
(CML-CP) previously treated
with 2 or more tyrosine kinase
inhibitors
20 NMRR-19-285- Dr Azizan Sharif Medical An Open-label, Long-term Safe- LTE 15174 Study start
46613 ty and Efficacy Study of Fitusir- up
an in Patients with Hemophilia
A or B, with or without Inhibitory
Antibodies to Factor VIII or IX
21 NMRR-19-770- Dr Lim Soo Min Medical A phase 3b, open-label, Believe Extension Study start
47233 single-arm, rollover study to up
evaluate long-term safety in
subjects who have participated
in other luspatercept (ace-536)
clinical trials
151